The flexible graphite sheet is widely used as sealing material in all kinds of industries,however,it has an advantage which is the metals in contact with the graphite usually get corrosion,and with the time goes by,this situation will be worse and worse.then the sealing effect will be failure,even cause a big accident.So we need to know the why this happen and where is the sulfur from?
Firstly ,we need to have a know on the production process of graphite sheet:
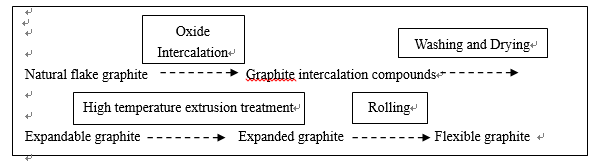
The raw material to make flexible graphite is natural flake graphite,in general,it is Fixed carbon with >99%,research shows that,the sulfur in the natural graphite is 200ppm below,far less than the flexible graphite (>1000ppm),then we could know that,the sulfur is not from the natural raw material,but comes from the production run.
The first step of the flexible graphite processing is Oxide Intercalation(chemical methods),and the most widely used intecalater is Concentrated sulfuric acid, massive sulfur element get into the graphite interlayer,after that,we could wash the graphite intercalation compounds and dry it,most acid is washed out,but still a portion remained(about 2.8%~4.5%).when the intermediate product is sent into high temperature extrusion treatment,the interlayer substance will be decomposition gasification(SO2、SO3、H2O etc.),and the gas will get into the furnace,the sulfur of the expanded graphite now is about 1000~2000ppm.after rolling the flexible graphite will be done.
Based on the analysis above,we could have a know that the sulfur is mainly from the Oxide Intercalation,other processing like washing,drying,extrusion and rolling,they could reduce the sulfur content.
So we could say that,the sulfur in the flexible graphite sheet is from Oxide Intercalation.